Analytical Advantages of SFC
The supercritical carbon dioxide used as a mobile phase for SFC offers unique advantages. Five of the advantages of using supercritical carbon dioxide for SFC analysis are described below.
- Low Polarity
The polarity of supercritical carbon dioxide is similar to n-hexane. - Miscibility with Other Organic Solvents
Supercritical carbon dioxide can be mixed with methanol, which is a water-soluble solvent. In contrast, n-hexane, which offers low polarity similar to supercritical carbon dioxide, cannot be mixed with methanol, due to its relatively high polarity. - Low Viscosity
Supercritical carbon dioxide has lower viscosity than solvents such as water. That results in lower column back pressure, assuming the column has the same internal diameter and length, is packed with the same size particles, and supercritical carbon dioxide is pumped through the column at the same rate. - Vaporization
Supercritical carbon dioxide evaporates at a temperature and pressure lower than the critical point. - Solvent Cost
Similar polarity of super critical CO2 is and of n-hexane, makes former appropriate to be used for normal phase separations. Normal-phase chromatography consumes large amounts of solvent, however SFC consumes less solvent and involves treating lesser liquid waste, the overall solvent cost is lower than for HPLC (Fig. 1).

Fig. 1 Comparison of Analytical Costs and Solvent Consumption Quantities for HPLC and SFC
The following sections describe the analytical advantages of SFC provided by the supercritical carbon dioxide advantages (1) to (4) indicated above.
Low Polarity – Better Structural Identification
Normal-phase chromatography is a separation method that uses a solvent with low-polarity, such as n-hexane, as the mobile phase and a substance with high-polarity, such as silica, as the stationary phase. One advantage of normal-phase chromatography is it offers better structure recognition ability than reversed-phase chromatography.
SFC uses supercritical carbon dioxide, which has similar properties as n-hexane, and also offers excellent structural recognition ability similar to normal-phase chromatography (Fig. 2). By using a column designed for chiral chromatography, which is packed with a stationary phase with optically active functional groups, SFC suitable for separation of chiral compounds.
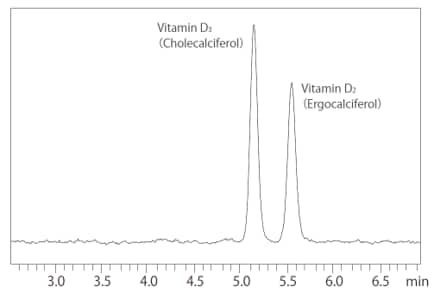
Fig. 2 Separating Vitamins D2 and D3 (UC-PyE Column)
Miscibility with Organic Solvents — Separations Covering Larger Chemical Space
Supercritical carbon dioxide can be mixed with certain organic solvents, such as methanol, ethanol, isopropyl alcohol (IPA), acetonitrile, and tetrahydrofuran (THF). By mixing the supercritical carbon dioxide with such modifiers, SFC can achieve a wide variety of separation patterns. This allows comprehensive analysis for a wide range of low-to-high polarity compounds not previously possible with HPLC. For example, earlier GC was required for analyzing fatty acids and HPLC for glycerides. With SFC, both fatty acids and glycerides can now be analyzed simultaneously due to the hexane-like properties of the supercritical carbon dioxide in combination with a column designed for reversed-phase chromatography (Fig. 3).
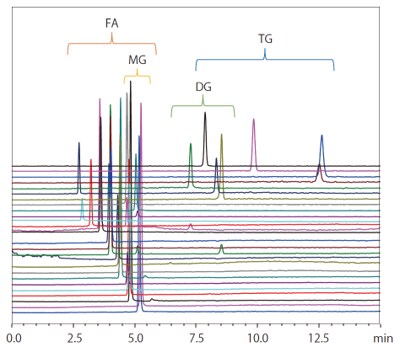
Fig. 3 Example of Simultaneously Analyzing Fatty Acids and Triglycerides by SFC
Low Viscosity — Achieving Ultra-Fast Analysis
Supercritical carbon dioxide has both low viscosity and a high diffusion coefficient. That enables analysis with a high linear velocity (flow speed with respect to the column cross-section). Fig. 4 shows a comparison of linear velocity and column pressure plots for HPLC and SFC.
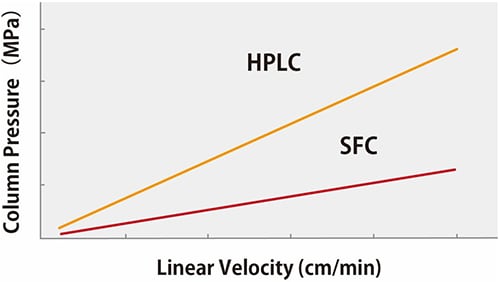
Fig. 4 Linear Velocity vs. Column Back Pressure
Though higher linear velocity can shorten HPLC analysis time, but it results in increased column pressure. Upper range of column pressure is limited by instrument design and column capability. CO2 in the supercritical state has lower viscosity than water and other organic solvents typically used for HPLC. That means shorter analysis time are possible for SFC than HPLC, without loss of resolution and separation pattern.
Supercritical carbon dioxide also has a higher coefficient of diffusion than solvents used for LC. The diffusion coefficient contributes to determining how quickly compounds reach equilibrium between stationary and mobile phases during chromatography and affects resolution. The van-Deemter equation below indicates the relationship between physical parameters that determine the height equivalent to a theoretical plate (HETP) value, which is used to indicate column resolution.

Plots of the van-Deemter equation for HPLC and SFC are shown in Fig. 5.*1 Term A is based on the multi-flow diffusion generated by the packing material in the column and is proportional to the packing material particle size. Term B is determined by the molecular diffusion in the axial direction of the column. This diffusion phenomenon occurs while compounds stagnate in the stationary or mobile phases and is inversely proportional to linear velocity, regardless of particle size. Term C is derived from how easily compounds can penetrate pores in the packing material and the mass transfer diffusion that contributes to linear velocity. The mass transfer diffusion value is proportional to linear velocity and the square of the particle diameter, which results in higher HETP values for small particles with low linear velocities. In the case of HPLC, mobile phases with a high linear velocity will result in larger mass transfer diffusion values, but supercritical carbon dioxide has a higher diffusion coefficient than HPLC solvents and can penetrate packing material pores more easily. That results in a lower mass transfer diffusion values even for high linear velocities.
That makes SFC an analytical technique that can achieve ultra-fast analysis without sacrificing resolution even at high linear velocities.
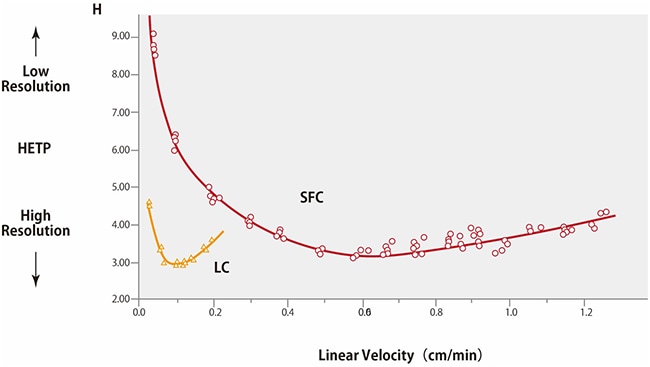
Fig. 5 Van-Deemter Plot Comparison for LC and SFC
These plots indicate the regions where the optimal linear velocity for SFC is higher than for HPLC. An example comparing the analysis of tocopherols by HPLC and SFC is shown in Fig. 6.
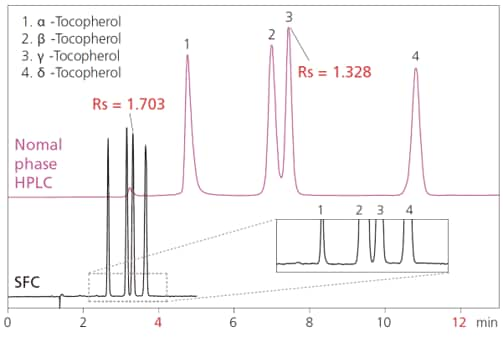
Fig. 6 Comparison Example of Analyzing Tocopherols by HPLC and SFC
Vaporization — Improved LC-MS Detection Sensitivity
Under certain temperature and pressure conditions, CO2 will become a solvent, which is used as a mobile phase for SFC. If an MS unit is used as a detector, then a back pressure regulator is installed just before the MS unit, so that only the modifier or make-up solvent reaches the MS unit. Consequently, unlike conventional LC-MS analysis, this reduces the diluting effects of the mobile phase, consequently potentially increasing the sensitivity whilst using SFC-MS. This can be seen from below mentioned example where 400 components were analyzed with 90% of these showing higher sensitivity with SFC-MS (Fig. 7). Fig. 8 shows a comparison of results from using LC-MS and SFC-MS to measure samples prepared to 10 ppb.
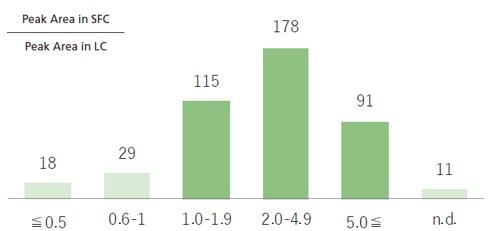
Fig. 7 Area Comparison for LC-MS and SFC-MS
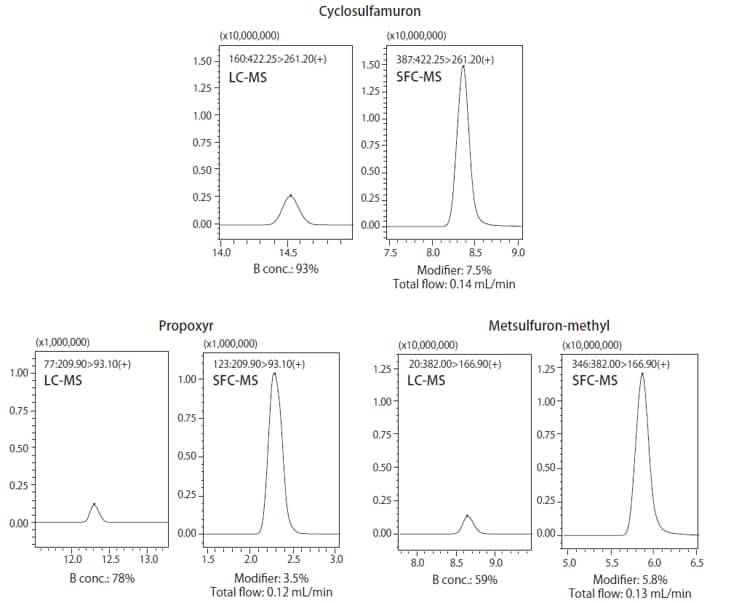
Fig. 8 Comparison of Mass Chromatograms of Standard Pesticide Mixture Solution Measured by LC-MS and SFC-MS
- The dependence of reduced plate height on reduced velocity in carbon dioxide supercritical fluid chromatography with packed columns Chromatographia 1987 Jan; 21-25






